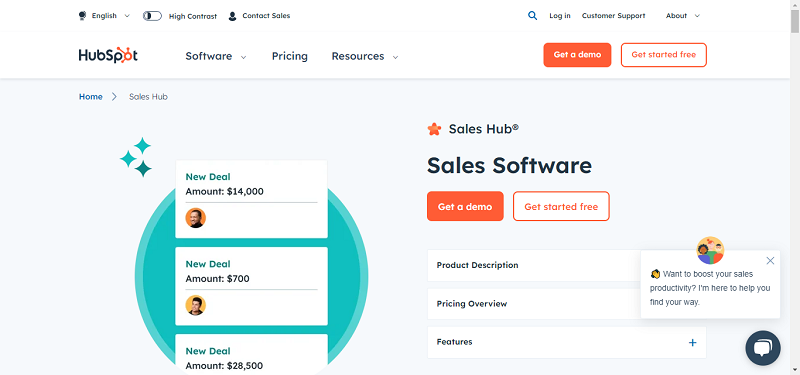
An individual in the South African trial of the AstraZeneca-Oxford COVID-19 vaccine has actually blood drawn prior to getting her 2nd dosage.
AP Photo/Jerome Hold-up.
Science‘s COVID-19 reporting is supported by the Heising-Simons Structure.
Another COVID-19 vaccine has actually encountered difficulty in South Africa, revealing less defense there than somewhere else since a SARS-CoV-2 variation that can obviously evade crucial antibodies has actually ended up being prevalent. In the wake of the brand-new finding, the nation, which had actually pinned its pandemic hopes on this specific vaccine, stopped an immunization project introduced just recently.
The stakes are high for the vaccine worldwide since its makers, AstraZeneca and the University of Oxford, hope it will be extensively utilized in establishing nations; they predict they can produce 3 billion dosages this year for around $3 each, even more item at a far less lower rate than any other vaccine revealed to provide defense versus COVID-19.
Associated
Yet the South African trial of the vaccine, carried out in about 2000 individuals, discovered such a low effectiveness versus moderate and moderate illness, under 25%, that it would not fulfill very little global requirements for emergency situation usage. However researchers are enthusiastic it may still avoid serious illness and death– probably the most crucial task for any COVID-19 vaccine. That was difficult to distinguish this placebo-controlled trial since it was little and hired reasonably healthy, youths– their typical age was just 31. None of the topics in either arm of the research study established serious illness or needed hospitalization.
The brand-new outcomes are a “truth check,” Shabir Mahdi of the University of Witwatersrand, the trial’s primary private investigator, stated today at an interview. “It is time regrettably for us to recalibrate our expectations of COVID-19 vaccines, in addition to how we tackle choosing how to react to the COVID-19 pandemic in South Africa in addition to worldwide.”
COVID-19 vaccines made by Johnson & & Johnson( J&J) and Novavax have actually likewise been revealed to provide weaker defense versus B. 1.351 (likewise called 501. V2), the SARS-CoV-2 variation that now triggers the huge bulk of all infections in South Africa, than versus older variations. The vaccines’ effectiveness versus moderate illness in South Africa was 57% for J&J and 49% for Novavax– lower than in any other nation they were checked.
However the J&J vaccine, which was tested in the biggest of the research studies, convincingly safeguarded versus serious illness and death, even versus the B. 1.351 alternative, and Mahdi stays “rather positive” that the AstraZeneca-Oxford vaccine will too; the outcomes are not “all doom and gloom,” he stated.
SARS-CoV-2-targeting antibodies set off by the J&J vaccine were “really comparable”, he stated, to those generated by the AstraZeneca-Oxford prospect, and the 2 vaccines are based upon a comparable innovation: Both cause the body to make the spike surface area protein of SARS-CoV-2 by providing its genes in a safe adenovirus. In a 44,000-person trial, the J&J vaccine avoided 85% of serious cases and entirely safeguarded individuals from hospitalization and death in numerous nations, consisting of the 15% of the individuals who were from South Africa.
Mahdi and the research study group had actually prepared to report the outcomes tomorrow, however the Financial Times on Saturday ran a story based upon a dripped copy of the findings. Mahdi and his colleagues have actually sent a paper explaining their information to a preprint server and anticipate it will publish tomorrow.
The AstraZeneca-Oxford vaccine has actually produced complicated arise from the start. Earlier initial arise from trials in various nations revealed a vast array of success rates versus moderate and moderate illness, however scientists have actually had problem analyzing the information since of distinctions in dosage, periods in between dosages, and variations in flow. Simply on Friday, a research study recommended the vaccine used strong defense versus a more transmissible variation, B. 1.1.7, that took off in the UK and is now spreading out quick throughout Europe.
In South Africa, the vaccine was given up 2 dosages spaced 21 to 35 days apart. Antibodies made by vaccine receivers can normally “reduce the effects of” SARS-CoV-2, indicating they can avoid it from contaminating cells in culture experiments. However laboratory research studies reveal that they have far less power versus B. 1.351.
Mahdi worried that the vaccine might still set off an effective T-cell reaction, which can target and remove cells the alternative handles to contaminate. He provided a test-tube research study demonstrating how the anomalies in the spike protein that permit B. 1.351. to evade reducing the effects of antibodies have little effect on T cell actions. “Our company believe that those T cell actions will still stay undamaged in spite of the anomalies that exists in a B. 1.351 alternative,” stated Mahdi.
The AstraZeneca-Oxford vaccine trial, which ranged from June to November, discovered that beginning 2 weeks after the 2nd dosage– when individuals probably were completely vaccinated– 19 cases of moderate or moderate illness established amongst the immunized, versus 23 in the placebo group, leading to an effectiveness of 21.9%. That is far listed below the 50% minimum needed for emergency situation usage permission in numerous nations.
Scientists sequenced the infections that contaminated trial individuals and discovered a strong link in between vaccine failure and B. 1.351’s surge in South Africa. In individuals who got one dosage of the vaccine prior to the alternative started to spread out extensively, effectiveness versus moderate and moderate illness was still a reputable 75%.
South Africa recently got 1 million dosages of the AstraZeneca-Oxford vaccine and started to provide them to health care employees, making it the very first COVID-19 vaccine readily available in the nation beyond medical trials. Epidemiologist Salim Abdool Karim, who co-chairs the South African Ministerial Advisory Committee on COVID-19, stated at journalism conference that the rollout of the vaccine in South Africa “requires to be placed on momentary hold” because of the frustrating outcomes. Barry Schoub, who leads a federal government advisory subcommittee on COVID-19 vaccines, states “we might require to take a look at mixes of the [AstraZeneca-Oxford] vaccine with other vaccines, which might in reality synergistically provide an excellent reaction.”
The University of Oxford group that initially developed the vaccine states it currently has actually started dealing with a second-generation prospect that targets the altered spike protein of the B. 1.351 variation. Oxford’s Sarah Gilbert, who leads that effort, recommended in a press declaration that a reformulated vaccine may be provided as a booster shot to the existing one.” This is the exact same problem dealt with by all of the vaccine designers, and we will continue to keep track of the development of brand-new variations that emerge in preparedness for a future stress modification,” kept in mind Gilbert.





/cdn.vox-cdn.com/uploads/chorus_asset/file/25405399/1247366679.jpg)